
Chemistry, 15.07.2020 21:01 duncanswart1558
A sample of Ne gas has a pressure of 654 mmHg with an unknown volume. The gas has a pressure of 345 mmHg when the volume is 495mL with no change in temperature or amount of gas. What is the initial volume in milliliters of the gas?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, sabahfayaskhan
Which of the following statements is true? question 4 options: nuclear decay rates vary with the conditions of the reaction, but chemical reaction rates do not. chemical reaction rates vary with the conditions of the reaction, but nuclear decay rates do not. neither chemical reaction rates nor nuclear decay rates vary with the conditions of the reaction. both chemical reaction rates and nuclear decay rates vary with the conditions of the reaction.
Answers: 1


Chemistry, 22.06.2019 12:30, emmalybrown
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1

Chemistry, 22.06.2019 18:20, juansebas35
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2
You know the right answer?
A sample of Ne gas has a pressure of 654 mmHg with an unknown volume. The gas has a pressure of 345...
Questions in other subjects:



History, 18.05.2021 19:00

Mathematics, 18.05.2021 19:00

History, 18.05.2021 19:00

Mathematics, 18.05.2021 19:00

History, 18.05.2021 19:00



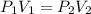
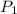 represent the initial pressure
represent the initial pressure represent the final pressure
represent the final pressure represent the initial temperature
represent the initial temperature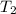 represent the final temperature
represent the final temperature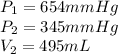




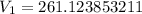
 (Approximated)
(Approximated)


