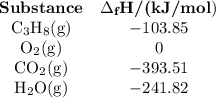Chemistry, 15.07.2020 20:01 mariakelley15
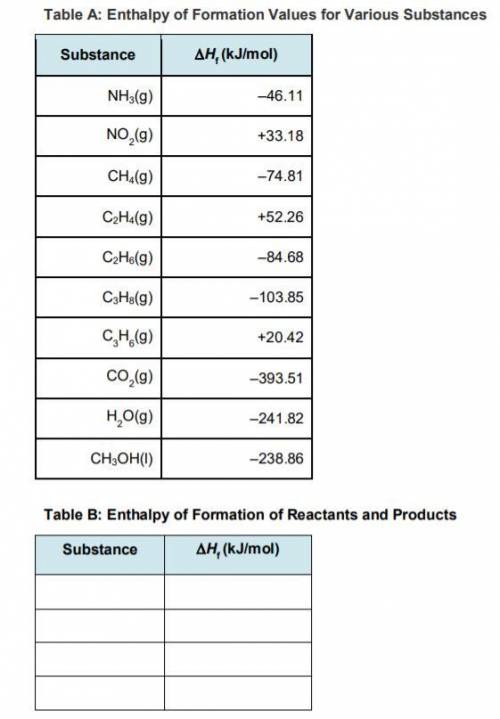
Step 3: Determine the amount of energy change in the reaction. a) Use the table of enthalpy values (Table A) provided in the Student Worksheet to locate the enthalpy of formation (∆Hf) for each reactant and each product. Record these values along with the reactants and products in Table B of the Student Worksheet. b) Determine the total enthalpy of the reactants and the total enthalpy of the products. Record these values in Table C of the Student Worksheet. c) Use the following formula to find the net change in enthalpy for the reaction and to determine whether the reaction is endothermic or exothermic.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, stelllllllllllllllla
George is a dalmatian puppy. describe what happens to light that allows you to see george’s black and white coat.
Answers: 1

Chemistry, 22.06.2019 17:10, gungamer720
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 22.06.2019 19:00, cindyroxana229
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3

Chemistry, 23.06.2019 00:30, tateandvioletAHS14AY
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
You know the right answer?
Step 3: Determine the amount of energy change in the reaction. a) Use the table of enthalpy values (...
Questions in other subjects:

Mathematics, 13.10.2020 01:01




Mathematics, 13.10.2020 01:01

History, 13.10.2020 01:01

Mathematics, 13.10.2020 01:01