
Chemistry, 14.07.2020 01:01 ondreabyes225pcr83r
Consider the following reaction where Kc = 6.50×10-3 at 298 K: 2NOBr(g) 2NO(g) + Br2(g) A reaction mixture was found to contain 9.83×10-2 moles of NOBr(g), 5.44×10-2 moles of NO(g), and 4.13×10-2 moles of Br2(g), in a 1.00 liter container. Is the reaction at equilibrium? If not, what direction must it run in order to reach equilibrium? The reaction quotient, Qc

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Garciaapril1597
An exothermic reaction is conducted in an insulated calorimeter filled with water. the calorimeter is then sealed so that there is no heat exchanged between the contents of the container and the surrounding air. which of the following statements is true about the reaction?
Answers: 1


Chemistry, 22.06.2019 14:40, sugardime
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2
You know the right answer?
Consider the following reaction where Kc = 6.50×10-3 at 298 K: 2NOBr(g) 2NO(g) + Br2(g) A reaction m...
Questions in other subjects:


Health, 30.07.2019 11:30

Arts, 30.07.2019 11:30

English, 30.07.2019 11:30



History, 30.07.2019 11:30

Mathematics, 30.07.2019 11:30

History, 30.07.2019 11:30

Mathematics, 30.07.2019 11:30

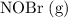 ) in order to reach an equilibrium.
) in order to reach an equilibrium.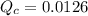 .
.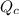 is equal to the equilibrium constant
is equal to the equilibrium constant 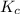 .
. 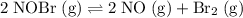 .
.![[\mathrm{NOBr\; (g)}]](/tpl/images/0704/6105/9e238.png) ,
, ![[\mathrm{NO\; (g)}]](/tpl/images/0704/6105/b4859.png) , and
, and ![[\mathrm{Br_2\; (g)}]](/tpl/images/0704/6105/fc290.png) denote the concentration of the three species. The formula for the reaction quotient of this system will be:
denote the concentration of the three species. The formula for the reaction quotient of this system will be:![\displaystyle Q_c = \frac{[\mathrm{NO\; (g)}]^2 \cdot [\mathrm{Br_2\; (g)}]}{[\mathrm{NOBr\; (g)}]^2}](/tpl/images/0704/6105/e21d0.png) .
.![\displaystyle Q_c = \frac{[\mathrm{NO\; (g)}]^2 \cdot [\mathrm{Br_2\; (g)}]}{[\mathrm{NOBr\; (g)}]^2} \approx 1.26\times 10^{-2} = 0.0126](/tpl/images/0704/6105/6c71a.png) .
.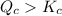 . Since
. Since 

