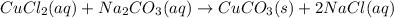Mixing which 0.1 M aqueous solutions results in formation of a colored precipitate? A) BaCl2 and CH3COOH (B) BaCl2 and Na2CO3 (C) CuCl2 and CH3COOH (D) CuCl2 and Na2CO3 The correct answer is D but please explain I'm confused. I know that the product has to be insoluble to form a precipitate, but how do you know if it'll be colored?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:40, gabrielolivas59
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3


Chemistry, 22.06.2019 21:30, emmalucilleblaha1995
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
Mixing which 0.1 M aqueous solutions results in formation of a colored precipitate? A) BaCl2 and CH3...
Questions in other subjects:


World Languages, 12.02.2022 03:40

English, 12.02.2022 03:40

Mathematics, 12.02.2022 03:40



Mathematics, 12.02.2022 03:50

History, 12.02.2022 03:50