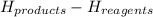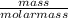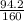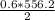A chemist measures the energy change Delta H during the following
2Fe2O3(s)->4FeO(s)+O2(g).
1) this reactions is: Endothermic or exothermic.
2) suppose 94.2g of Fe2O3 react. will any heat be relased or absorbed. yes absorbed. yes releases. no.
3) If you said heat will be released or absorbed in the second part of the question. calculate how much heat will be absored or released. be sure your answer has correct number of significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, angelrenee2000
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 18:00, ameliaxbowen7
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1

Chemistry, 22.06.2019 21:00, rhondafits9000
Which property of water causes water drops to bead on a freshly waxed car?
Answers: 2
You know the right answer?
A chemist measures the energy change Delta H during the following
2Fe2O3(s)->4FeO(s)+O2(g).
Questions in other subjects:







Advanced Placement (AP), 26.12.2019 05:31

Spanish, 26.12.2019 05:31

Chemistry, 26.12.2019 05:31