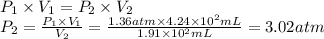Chemistry, 06.07.2020 17:01 lyssamichelle
A sample of oxygen, at 25°C, occupies a volume of 4.24 × 102 milliliters (mL) at 1.36 atm pressure. What pressure must be applied to compress the gas to a volume of 1.91 × 102 mL, with no temperature change?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, jbarbie3
12. complete each of the following word equations for synthesis reactions. a. sodium + oxygen -> b. magnesium + fluorine -> 13. complete and balance the equations for the decomposition reactions. a. hgo -> [with the triangle heat symbol above the arrow] b. h2o(l) -> [with "electricity" written above the arrow]
Answers: 1


Chemistry, 23.06.2019 09:00, notearslefttocry14
What sources of error may have contributed to the percent yield not being 100 percent? think about things that may have led to inaccurate measurements or where mass of the product could have been lost if this experiment was conducted in a physical laboratory.
Answers: 2

Chemistry, 23.06.2019 09:20, taylorannsalazar
La reaccion entre monoxido de nitrogeno (no) y oxigeno para formardioxido de nitrogeno (no2) es un paso determinante para la formacion del smog, la reaccion es la siguiente: 2no + o2 = 2no2 cual sera el numero de moles de no2 que se formaran por la reaccion completa de 8 moles de oxigeno con suficiente monoxido?
Answers: 1
You know the right answer?
A sample of oxygen, at 25°C, occupies a volume of 4.24 × 102 milliliters (mL) at 1.36 atm pressure....
Questions in other subjects:


Advanced Placement (AP), 30.09.2019 19:20








History, 30.09.2019 19:20