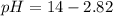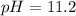Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:00, kcarstensen59070
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 09:40, loveoneonly9153
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
You know the right answer?
A chemist dissolves 867. mg of pure barium hydroxide in enough water to make up 170. mL of solution....
Questions in other subjects:


Mathematics, 02.04.2021 23:50

Mathematics, 02.04.2021 23:50





Mathematics, 02.04.2021 23:50

English, 02.04.2021 23:50

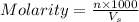
 = volume of solution in ml
= volume of solution in ml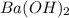 =
= 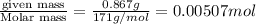 (1g=1000mg)
(1g=1000mg)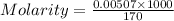
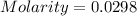
![pOH=-\log [OH^-]](/tpl/images/0701/6467/1fac1.png)
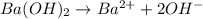
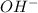
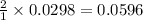 moles of
moles of ![pOH=-\log[0.0596]=2.82](/tpl/images/0701/6467/015aa.png)