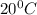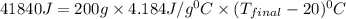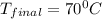Chemistry, 03.07.2020 22:01 Nashae4771
If 200. g of water at 20°C absorbs 41 840 J of energy, what will its final temperature be? (Specific Heat of water is 4.184 J/g*C)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1


Chemistry, 23.06.2019 03:30, tamariarodrigiez
Which of the following describes the entropy change as a solution is made from a liquid and solid
Answers: 1

Chemistry, 23.06.2019 05:40, Queenquestion9130
Why is any chemical reaction always balanced? give reasons and explain the easiest way to solve the balancing problems in chemical equations with stoichiometric coefficients upto 20 as hit and trial doesn't always work. give full reasoning
Answers: 1
You know the right answer?
If 200. g of water at 20°C absorbs 41 840 J of energy, what will its final temperature be? (Specific...
Questions in other subjects:




Mathematics, 10.11.2019 23:31


Computers and Technology, 10.11.2019 23:31


Mathematics, 10.11.2019 23:31

Mathematics, 10.11.2019 23:31

Mathematics, 10.11.2019 23:31

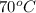
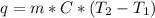
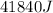
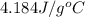
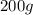
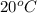
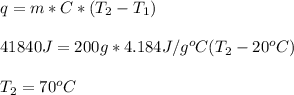
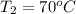

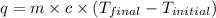
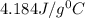
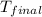 = final temperature =?
= final temperature =?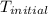 = initial temperature =
= initial temperature =