Chemistry, 03.07.2020 22:01 andybiersack154
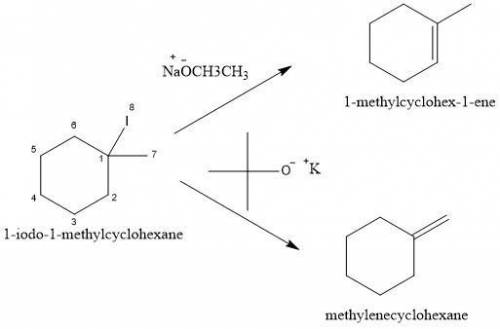
When 1-iodo-1-methylcyclohexane is treated with NaOCH2CH3 as the base, the more highly substituted alkene product predominates. When KOC(CH3)3 is used as the base, the less highly substituted alkene predominates. Give the structures of the two products and offer an explanation.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:20, kingaman
Which of the following statements is not true? • a. covalent compounds have low melting and boiling points. • ob. covalent bonds between atoms of a compound are relatively weak compared to bonds between molecules. • c. covalent bonds occur between nonmetals. • d. covalent compounds are often gases or liquids.
Answers: 2

Chemistry, 21.06.2019 15:30, officialrogerfp3gf2s
How to solve 4 nh3(g) + 5 o2(g) > 4 no(g) + 6 h2o(g) in chemistry
Answers: 1

Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

You know the right answer?
When 1-iodo-1-methylcyclohexane is treated with NaOCH2CH3 as the base, the more highly substituted a...
Questions in other subjects:

Chemistry, 01.08.2019 11:00

History, 01.08.2019 11:00

Social Studies, 01.08.2019 11:00


Mathematics, 01.08.2019 11:00


Biology, 01.08.2019 11:00


Health, 01.08.2019 11:00

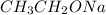 is a strong base but only has 2 carbons therefore we will have less steric hindrance in this base. So, the base can remove hydrogens that are bonded on carbons 1 or 6, therefore, we will have a more substituted alkene (1-methylcyclohex-1-ene).
is a strong base but only has 2 carbons therefore we will have less steric hindrance in this base. So, the base can remove hydrogens that are bonded on carbons 1 or 6, therefore, we will have a more substituted alkene (1-methylcyclohex-1-ene).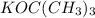 we have more steric hindrance. So, we can remove only the hydrogens from carbon 7 and we will produce a less substituted alkene (methylenecyclohexane).
we have more steric hindrance. So, we can remove only the hydrogens from carbon 7 and we will produce a less substituted alkene (methylenecyclohexane).