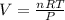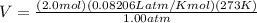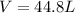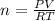Chemistry, 02.07.2020 03:01 goalltheway4768
A closed container holds 2.0 moles of CO2 gas at STP. How many moles of oxygen can be placed in a container of the same size at STP?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:30, latezwardjr15
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2

Chemistry, 22.06.2019 20:30, Schoolworkspace453
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 23.06.2019 00:30, hdhshshs741
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1
You know the right answer?
A closed container holds 2.0 moles of CO2 gas at STP. How many moles of oxygen can be placed in a co...
Questions in other subjects:


Mathematics, 29.06.2019 18:30



Chemistry, 29.06.2019 18:30

Geography, 29.06.2019 18:30



History, 29.06.2019 18:30

Chemistry, 29.06.2019 18:30