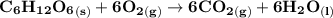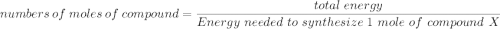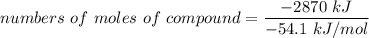Chemistry, 02.07.2020 22:01 treypickich14
Assume that the complete combustion of one mole of glucose, a monosaccharide, to carbon dioxide and water liberates 2870 kJ2870 kJ of energy (ΔG°′=−2870 kJ/mol(ΔG°′=−2870 kJ/mol ). If the energy generated by the combustion of glucose is entirely converted to the synthesis of a hypothetical compound X, calculate the number of moles of the compound that could theoretically be generated. Use the value ΔG°′compound X=−54.1 kJ/molΔG°′compound X=−54.1 kJ/mol . Round your answer to two significant figures.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, villarrealc1987
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2


Chemistry, 22.06.2019 19:30, amandamiro05
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2
You know the right answer?
Assume that the complete combustion of one mole of glucose, a monosaccharide, to carbon dioxide and...
Questions in other subjects:



Mathematics, 19.07.2019 08:10

Mathematics, 19.07.2019 08:10

Health, 19.07.2019 08:10

English, 19.07.2019 08:10

Mathematics, 19.07.2019 08:10



 53 mole
53 mole