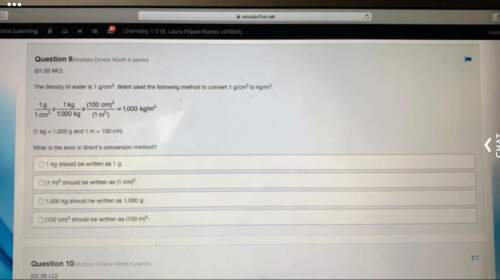
Chemistry, 02.07.2020 01:01 Rachelmontes1
How many electrons are exchanged in the following reaction? 2 Fe3+ (aq) + 3 Cu(s) = 2 Fe(s) + 3 Cu2+(aq)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, sammiehammer
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 21.06.2019 22:30, Aidanjsauer
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

You know the right answer?
How many electrons are exchanged in the following reaction?
2 Fe3+ (aq) + 3 Cu(s) = 2 Fe(s) + 3 Cu2...
Questions in other subjects:

Mathematics, 28.12.2020 20:20




Social Studies, 28.12.2020 20:20

World Languages, 28.12.2020 20:20

English, 28.12.2020 20:20


Mathematics, 28.12.2020 20:20

English, 28.12.2020 20:20