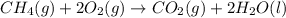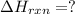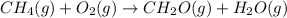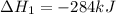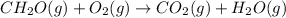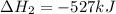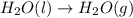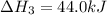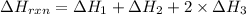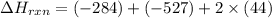Chemistry, 01.07.2020 22:01 catsRlife4573
Calculate ΔHrxnΔHrxn for the following reaction: CH4(g)+2O2(g)→CO2(g)+2H2O(l)CH4(g)+ 2O2(g)→CO2(g)+2H2O(l) Use the following reactions and given ΔHΔH values. CH4(g)+O2(g)→CH2O(g)+H2O(g)CH4(g)+O 2(g)→CH2O(g)+H2O(g), ΔH=−ΔH=−284 kJkJ CH2O(g)+O2(g)→CO2(g)+H2O(g)CH2O(g)+ O2(g)→CO2(g)+H2O(g), ΔH=−ΔH=−527 kJkJ H2O(l)→H2O(g)H2O(l)→H2O(g), ΔH=ΔH= 44.0 kJ

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, gilbert325
Write the chemical symbols for three different atoms or atomic cations with 27 electrons. asap!
Answers: 2

Chemistry, 22.06.2019 15:30, 20cschultz
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 16:00, jrocklove7825
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2
You know the right answer?
Calculate ΔHrxnΔHrxn for the following reaction: CH4(g)+2O2(g)→CO2(g)+2H2O(l)CH4(g)+ 2O2(g)→CO2(g)+2...
Questions in other subjects:

Biology, 09.12.2020 08:40

Biology, 09.12.2020 08:40





History, 09.12.2020 08:40

Mathematics, 09.12.2020 08:40

Chemistry, 09.12.2020 08:40

Business, 09.12.2020 08:40