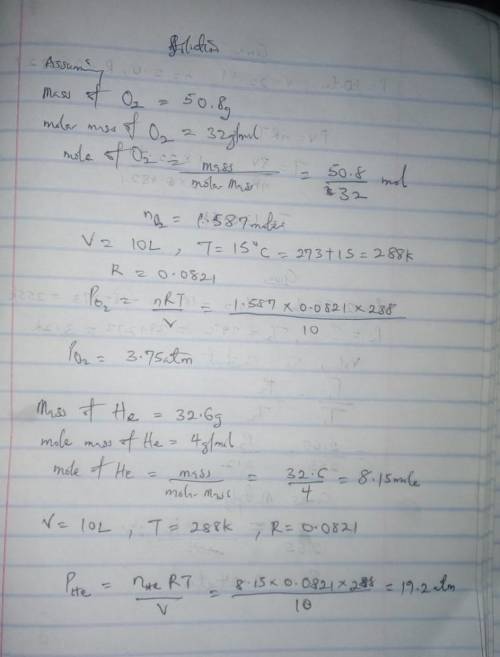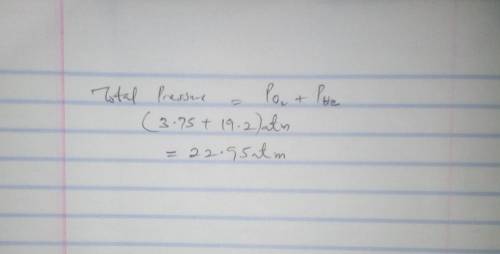Chemistry, 01.07.2020 16:01 rebekahwirogo
Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 15 ∘C.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 12:30, meghan2529
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1

Chemistry, 22.06.2019 12:30, poopybutt541
Avariable that is not being directly tested during an experiment should be
Answers: 1
You know the right answer?
Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 1...
Questions in other subjects:






Chemistry, 04.11.2019 05:31


Mathematics, 04.11.2019 05:31

Mathematics, 04.11.2019 05:31