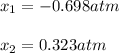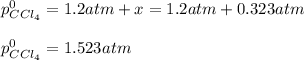Chemistry, 01.07.2020 15:01 diegobebe503
At 700 K, CCl4 decomposes to carbon and chlorine. The Kp for the decomposition is 0.76. Find the starting pressure of CCl4 at this temperature that will produce a total pressure of 1.2 atm at equilibrium. P=?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, bartfrank447
Joseph has hypothesized that sound travels in waves. if he were following the scientific method, what should he do next? a. ask a question. b. test the hypothesis. c. study the results. d. tell other scientists about his hypothesis.
Answers: 1

Chemistry, 22.06.2019 00:00, lukeakalucas
Alarge marble is dropped in a graduated cylinder with 35ml of water in it. the water level increases to 49ml. what is the volume of the marble
Answers: 1

Chemistry, 22.06.2019 11:00, bigwaYne
Imagine that twenty i. u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 12:30, americanbellabeauty
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1
You know the right answer?
At 700 K, CCl4 decomposes to carbon and chlorine. The Kp for the decomposition is 0.76. Find the sta...
Questions in other subjects:



Mathematics, 12.12.2020 16:20




History, 12.12.2020 16:20

Mathematics, 12.12.2020 16:20


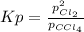
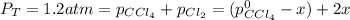
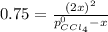
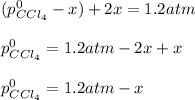
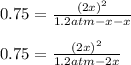
 we have to roots:
we have to roots: