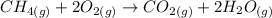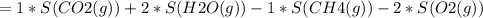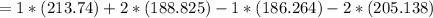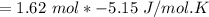Chemistry, 27.06.2020 18:01 destiny3200
Consider the reaction CH4(g) 2O2(g)CO2(g) 2H2O(g) Using standard thermodynamic data at 298K, calculate the entropy change for the surroundings when 1.62 moles of CH4(g) react at standard conditions.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, daniellekennedy05
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 12:00, macylen3900
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2


Chemistry, 22.06.2019 20:30, dinapaul424
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1
You know the right answer?
Consider the reaction CH4(g) 2O2(g)CO2(g) 2H2O(g) Using standard thermodynamic data at 298K, calcula...
Questions in other subjects:

Mathematics, 12.06.2020 01:57


History, 12.06.2020 01:57

Biology, 12.06.2020 01:57

Mathematics, 12.06.2020 01:57

Mathematics, 12.06.2020 01:57

Computers and Technology, 12.06.2020 01:57

Mathematics, 12.06.2020 01:57


Mathematics, 12.06.2020 01:57