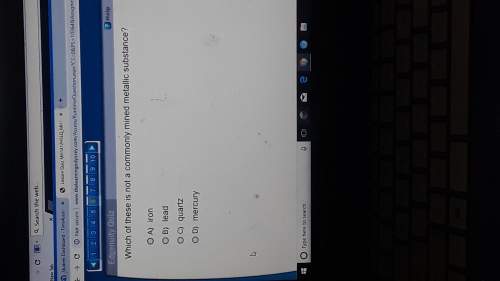
Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, earcake2470
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 06:00, giusto1894
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 23.06.2019 04:30, Har13526574
Two liquids are poured into a beaker. after a few seconds, the beaker becomes warm. which of the following best describes this reaction? a. an exothermic reaction b. a decomposition reaction c. an endothermic reaction d. a single-displacement reaction
Answers: 1

Chemistry, 23.06.2019 06:20, Lindsay882
Type the correct answer in each box. balance the chemical equation.__ n203 ➡️ __ n2 +__ o2
Answers: 1
You know the right answer?
You wish to make a 0.289 M hydroiodic acid solution from a stock solution of 3.00 M hydroiodic acid....
Questions in other subjects:

Mathematics, 08.09.2021 23:00

English, 08.09.2021 23:00



English, 08.09.2021 23:00



Mathematics, 08.09.2021 23:00

History, 08.09.2021 23:00

Physics, 08.09.2021 23:00

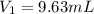


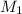 is the initial molarity (3.00 M) of the stock solution,
is the initial molarity (3.00 M) of the stock solution,  the molarity of the diluted solution (0.289 M),
the molarity of the diluted solution (0.289 M),  the aliquot of the stock (concentrated) solution and
the aliquot of the stock (concentrated) solution and 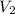 the volume of the diluted solution (100 mL), thus, we compute
the volume of the diluted solution (100 mL), thus, we compute