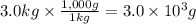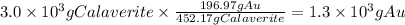Chemistry, 26.06.2020 16:01 jazminemendezhidalgo
What is the maximum mass of pure gold that could be extracted from 3.0kg of calaverite, a gold ore with the chemical formula AuTe2? Be sure your answer has a unit symbol, if necessary, and is rounded to 2 significant digits.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:30, huangjianhe135
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 23.06.2019 02:00, FailingstudentXD
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1

Chemistry, 23.06.2019 08:30, mhurtado143
This has nothing to do with school. i wrote a poem to my crush, who i'm asking out soon. tell me if it's cheesy, or cute. "roses are red, violets are blue no love story sounds right if it doesn't include you. dance with me all night, gaze into my eyes i'll hand you my heart, as well as my pride. when i hear your name, my heart goes insane. your all that i want, all that i need promise me you'll stay with me. here it is the final line, jasmine hill will you be mine? " i'm also going to buy her flowers, teddy bear and some food lol. written by me, bre (:
Answers: 2

Chemistry, 23.06.2019 10:30, cjtambasco
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 3.75 mol fe and 8.70 mol nio(oh) react?
Answers: 1
You know the right answer?
What is the maximum mass of pure gold that could be extracted from 3.0kg of calaverite, a gold ore w...
Questions in other subjects:


Mathematics, 05.07.2019 05:30


Physics, 05.07.2019 05:30

English, 05.07.2019 05:30

Biology, 05.07.2019 05:30




Spanish, 05.07.2019 05:30