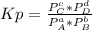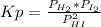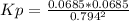Chemistry, 26.06.2020 15:01 wrightstephanie193
2. A reaction vessel is charged with hydrogen iodide, which partially decomposes to molecular hydrogen and iodine:2HI (g) H2(g) + I2(g)When the system comes to equilibrium at 425 °C, PHI = 0.794 atm, and PH2 = PI2 = 0.0685 atm. The value of Kp at this temperature is .

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:10, vapelordcarl69
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3


Chemistry, 23.06.2019 00:20, HernanJe6
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
2. A reaction vessel is charged with hydrogen iodide, which partially decomposes to molecular hydrog...
Questions in other subjects:



Business, 05.01.2020 06:31



History, 05.01.2020 06:31

Mathematics, 05.01.2020 06:31


History, 05.01.2020 06:31

Biology, 05.01.2020 06:31