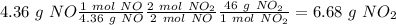Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 4.36 grams of nitrogen monoxide are mixed with excess oxygen gas . The reaction yields 5.46 grams of nitrogen dioxide . nitrogen monoxide ( g ) oxygen ( g ) nitrogen dioxide ( g ) What is the theoretical yield of nitrogen dioxide

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, NatalieKnows
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1



Chemistry, 23.06.2019 04:00, winterblanco
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 1
You know the right answer?
Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 4.3...
Questions in other subjects:

Arts, 30.11.2020 02:30


History, 30.11.2020 02:30

Mathematics, 30.11.2020 02:30



Mathematics, 30.11.2020 02:30


English, 30.11.2020 02:30

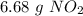

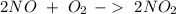
 . To do this, we have to first convert the 4.36 g of
. To do this, we have to first convert the 4.36 g of  to moles
to moles