Question 1:
Consider the nucleophilic substitution reaction between 1-chloro-
3,3-dimethylpen...

Chemistry, 23.06.2020 10:57 madmatt873
Question 1:
Consider the nucleophilic substitution reaction between 1-chloro-
3,3-dimethylpentane and the cyanide ion:
xa
+
ci
NC-
1-chloro-3,3-dimethylpentane
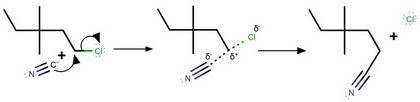
(a) Is the alkyl halide 1°, 2° or 3°?
(b) Will the nucleophilic substitution reaction be Sn1 or Sn2?
(c) Give the substitution mechanism showing all full and partial
positive and negative charges, electron movements using curved
arrows and any intermediates or transition states.
2

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, lorenaandreahjimenez
The answer for #3 is c but i don't know why
Answers: 1


Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
Questions in other subjects:

Mathematics, 28.07.2021 14:00

English, 28.07.2021 14:00

Computers and Technology, 28.07.2021 14:00


Arts, 28.07.2021 14:00

Mathematics, 28.07.2021 14:00

Mathematics, 28.07.2021 14:00



Mathematics, 28.07.2021 14:00