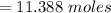Chemistry, 20.06.2020 20:57 tddreviews
A flexible container at an initial volume of 4.11 L contains 6.51 mol of gas. More gas is then added to the container until it reaches a final volume of 11.3 L. Assuming the pressure and temperature of the gas remain constant, calculate the number of moles of gas added to the container.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 04:00, Mitchmorgan3816
Why must humans find substitutes for many minerals found on earth? (a) form at an extremely slow rate (b) controlled by other countries (c) too deep in the earth to collect
Answers: 1

Chemistry, 23.06.2019 06:30, ayoismeisjjjjuan
What is the chemical formula for a compound between li and br? libr li2br libr2 libr3
Answers: 1
You know the right answer?
A flexible container at an initial volume of 4.11 L contains 6.51 mol of gas. More gas is then added...
Questions in other subjects: