
Chemistry, 27.09.2019 06:30 niyyyareligion
If 5 moles of p4 reacted with 22 moles cl2 according to the above reaction, determine:
a) how many moles pcl3 are produced
b) how many moles of p4 are left in excess after the reaction (if any)
c) how many moles of cl2 are left in excess after the reaction (if any)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, dustinsampsin2486
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1


Chemistry, 22.06.2019 17:50, kaylamount
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2
You know the right answer?
If 5 moles of p4 reacted with 22 moles cl2 according to the above reaction, determine:
a) how...
a) how...
Questions in other subjects:


History, 01.10.2019 19:40




Mathematics, 01.10.2019 19:50

Social Studies, 01.10.2019 19:50

Mathematics, 01.10.2019 19:50

Mathematics, 01.10.2019 19:50

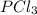 = 2.44 moles
= 2.44 moles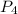 left = 1.34 mole
left = 1.34 mole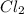 because it is completely consumed in the reaction.
because it is completely consumed in the reaction.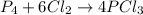
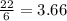 moles of
moles of 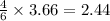 moles of
moles of 

