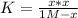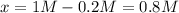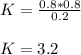Chemistry, 18.06.2020 05:57 andregijoe41
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed in one liter of solution. When equilibrium was established, 0.2 mole of PCl5 remained in the mixture. What is the equilibrium constant for this reaction? (Hint: remember the ICE procedure? initial, change, and equilibrium)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, Kjswagout5052
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 17:40, adantrujillo1234
Areaction in which products can react to re-form reactants is
Answers: 1


Chemistry, 22.06.2019 20:30, camerondillonn
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed i...
Questions in other subjects:



Mathematics, 12.05.2021 02:10

History, 12.05.2021 02:10

Arts, 12.05.2021 02:10


Mathematics, 12.05.2021 02:10



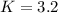
![K=\frac{[PCl_3][Cl_2]}{[PCl_5]}](/tpl/images/0688/6864/bc353.png)
 due to the reaction extent (ICE procedure):
due to the reaction extent (ICE procedure):