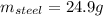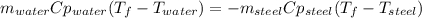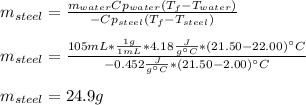Chemistry, 18.06.2020 02:57 Jessietorres2123
A volume of 105 mL of H2O is initially at room temperature (22.00 ∘C). A chilled steel rod at 2.00 ∘C is placed in the water. If the final temperature of the system is 21.50 ∘C , what is the mass of the steel bar? Use the following values: specific heat of water = 4.18 J/(g⋅∘C) specific heat of steel = 0.452 J/(g⋅∘C) Express your answer to three significant figures and include the appropriate units.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, ayoismeisalex
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 19:20, halledoll2002
Anyone who's in connections academy chemistry b have the factors that affect the rate of a reaction portfolio already done?
Answers: 3

Chemistry, 23.06.2019 00:00, PineappleDevil889
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 01:30, sheldonwaid4278
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1
You know the right answer?
A volume of 105 mL of H2O is initially at room temperature (22.00 ∘C). A chilled steel rod at 2.00 ∘...
Questions in other subjects:

Mathematics, 23.02.2021 17:10


Mathematics, 23.02.2021 17:10

Mathematics, 23.02.2021 17:10

Mathematics, 23.02.2021 17:10

Mathematics, 23.02.2021 17:10




Mathematics, 23.02.2021 17:10