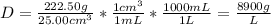Chemistry, 14.06.2020 22:57 mahmudabiazp3ekot
Use the mass and volume data to calculate the density of an unknown metal to the nearest hundredth mass of unknown metal = 222.50g volume of unknown metal = 25.00cm3 what is the density of the unknown metal

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, kaliyab191
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 20:00, aksambo4707
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 23.06.2019 01:00, aliviadushane
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1

Chemistry, 23.06.2019 02:00, FailingstudentXD
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Use the mass and volume data to calculate the density of an unknown metal to the nearest hundredth m...
Questions in other subjects:

Mathematics, 27.10.2020 17:00

Computers and Technology, 27.10.2020 17:00

Mathematics, 27.10.2020 17:00





Mathematics, 27.10.2020 17:00