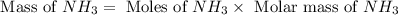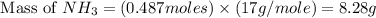Chemistry, 13.06.2020 14:57 snlawson9053
1.46 g H2 is allowed to react with 10.5 g N2, producing 2.72 g NH3. What is the theoretical yield in grams for this reaction under the given conditions? Express your answer to three significant figures and include the appropriate units.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, ggdvj9gggsc
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1


Chemistry, 22.06.2019 21:20, 50057543
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 22.06.2019 22:00, luciaaviles3
Pls ill give u brainliest which of the following is true about science? 1. political conditions are unable to influence it. 2. economic concerns may prevent it from solving problems.
Answers: 2
You know the right answer?
1.46 g H2 is allowed to react with 10.5 g N2, producing 2.72 g NH3. What is the theoretical yield in...
Questions in other subjects:

Advanced Placement (AP), 23.09.2019 19:10

Biology, 23.09.2019 19:10






Health, 23.09.2019 19:10


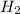 = 1.46 g
= 1.46 g
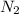 = 10.5 g
= 10.5 g
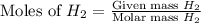
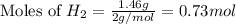
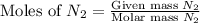
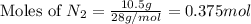
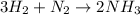
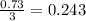 moles of
moles of 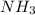
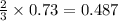 mole of
mole of