What would happen if the amount of sodium azide used was far greater or far less than what you calculated in part C? Describe both cases.
this was part C
2NaN3 → 2Na + 3N2.
Calculate the mass of sodium azide required to decompose and produce the number of moles of nitrogen you calculated in part B of this task. Refer to the periodic table to get the atomic weights.

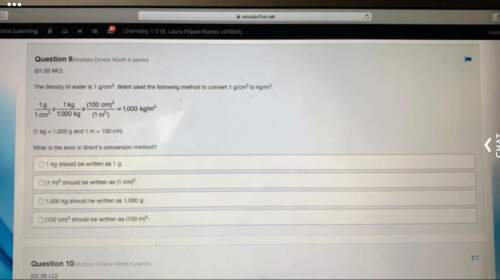
Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, lylessd423
Two atoms interact with each other as shown by the equation. complete the equation by filling in the missing parts. 1 2 3 4 5 h he li
Answers: 2

Chemistry, 22.06.2019 06:30, dimondqueen511
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1


Chemistry, 22.06.2019 14:00, hannahhoskings6989
What was bohr’s contribution to the planetary model
Answers: 1
You know the right answer?
What would happen if the amount of sodium azide used was far greater or far less than what you calcu...
Questions in other subjects: