
Chemistry, 12.06.2020 02:57 eddsworldfrantic
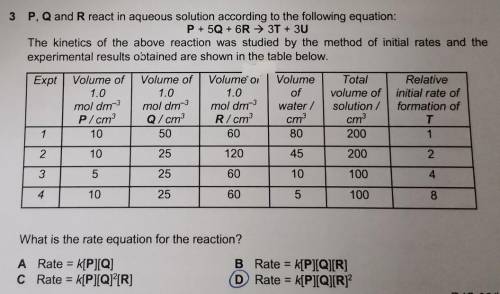
P, Q and R react in aqueous solution according to the following equation:
P + 50 + 6R → 3T + 3U
The kinetics of the above reaction was studied by the method of initial rates and the
experimental results obtained are shown in the table below
MCQ question (reaction kinetics)

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, jivsf
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
P, Q and R react in aqueous solution according to the following equation:
P + 50 + 6R → 3T + 3U
Questions in other subjects:

Geography, 24.07.2019 01:30


Social Studies, 24.07.2019 01:30

Biology, 24.07.2019 01:30




Spanish, 24.07.2019 01:30




