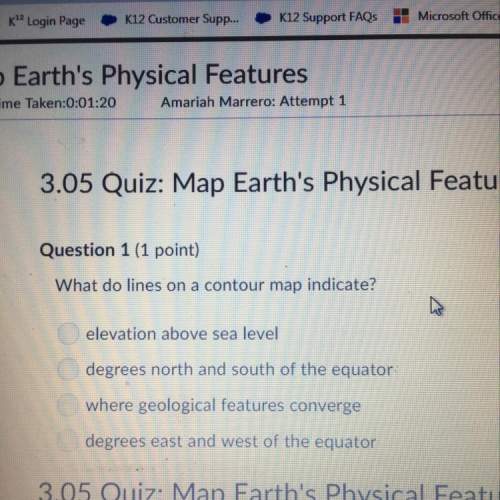
Chemistry, 06.06.2020 05:00 oranjejuice
Which of the following samples contains the greatest number of atoms?
a. 9 mole of CO2 b. 10 moles of Xe
c. 11 moles of N2O d. 12 moles of CO

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1
You know the right answer?
Which of the following samples contains the greatest number of atoms?
a. 9 mole of CO2 b. 10 moles...
Questions in other subjects:

Business, 30.01.2022 04:50

Mathematics, 30.01.2022 04:50

Biology, 30.01.2022 04:50

Chemistry, 30.01.2022 04:50

Mathematics, 30.01.2022 04:50

Law, 30.01.2022 05:00

Mathematics, 30.01.2022 05:00

Mathematics, 30.01.2022 05:00

History, 30.01.2022 05:00