Chemistry, 06.06.2020 03:57 cooljazzy1234
43. Calculate the equilibrium constant at the temperature given. (a) O2 (g) + 2F2 (g) ⟶ 2F2 O(g) (T = 100 °C) (b) I2 (s) + Br2 (l) ⟶ 2IBr(g) (T = 0.0 °C) (c) 2LiOH(s) + CO2 (g) ⟶ Li2CO3 (s) + H2 O(g) (T = 575 °C) (d) N2 O3 (g) ⟶ NO(g) + NO2 (g) (T = −10.0 °C) (e) SnCl4 (l) ⟶ SnCl4 (g) (T = 200 °C)

Answers: 2


Other questions on the subject: Chemistry




Chemistry, 23.06.2019 01:20, michellectucker1982
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
43. Calculate the equilibrium constant at the temperature given. (a) O2 (g) + 2F2 (g) ⟶ 2F2 O(g) (T...
Questions in other subjects:

Mathematics, 24.07.2019 18:00

Physics, 24.07.2019 18:00







Health, 24.07.2019 18:00

History, 24.07.2019 18:00

![\Delta G_f[F_2O]=41.9kJ/mol =41900J/mol](/tpl/images/0678/2670/ad240.png)
![\Delta G_f[O_2]=0\\\\ \Delta G_f[F_2]=0](/tpl/images/0678/2670/20ae9.png)
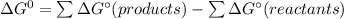
![\Delta G^\circ = [2 \times 41900]-0\\\\=83800J/mol](/tpl/images/0678/2670/d8ac8.png)