Chemistry, 05.06.2020 10:57 demetricejames
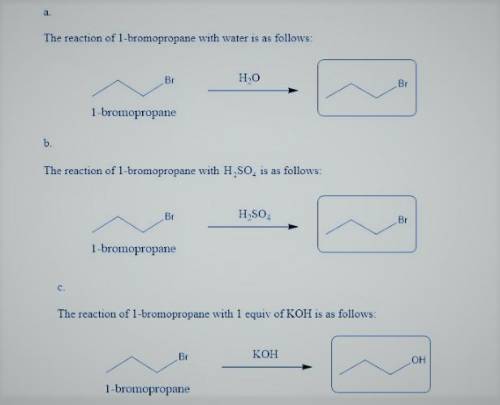
1-Bromopropane is treated with each of the following reagents. Draw the major substitution product if the reaction proceeds in good yield. If no reaction is expected or yields will be poor, draw the starting material in the box. If a charged product is formed, be sure to draw the counterion.
a. with H_2 O
b. with H_2 SO_4
c. with 1 equiv of KOH
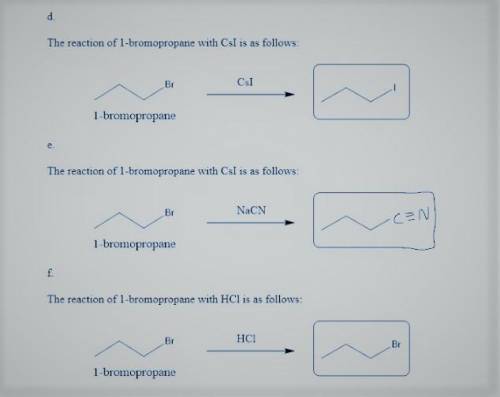
d. with Csl
e. with NaCN
f. with HCl
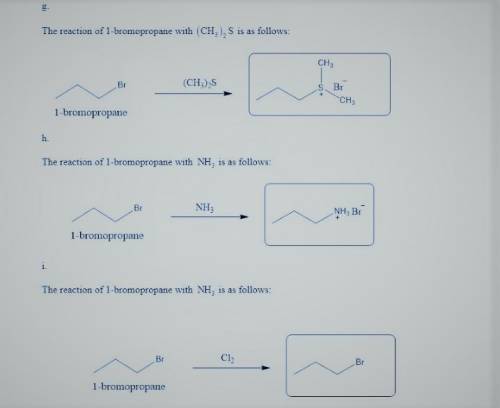
g. with (CH_3)_2 S
h. with 1 equiv of NH_3
j. with CI_2
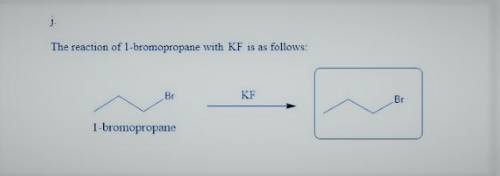
k. with KF

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 06:30, noathequeen
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 19:00, cindyroxana229
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3

Chemistry, 23.06.2019 07:20, msladycie8831
F1.5 mol of nabh4 react, how many moles of b2h6 are formed? 2 nabh4(aq) + h2so4(aq) → 2 h2(g) + na2so4(aq) + b2h6(g)
Answers: 1
You know the right answer?
1-Bromopropane is treated with each of the following reagents. Draw the major substitution product i...
Questions in other subjects:

Mathematics, 12.02.2020 19:51

Social Studies, 12.02.2020 19:51

English, 12.02.2020 19:51




Health, 12.02.2020 19:51

Social Studies, 12.02.2020 19:51

Mathematics, 12.02.2020 19:51