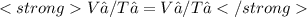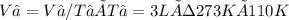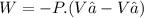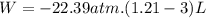Chemistry, 05.06.2020 00:00 SweetBriar
We now consider three moles of ideal gas at the same initial state (3.0 L at 273 K). This time, we will first perform an isobaric compression, then an isothermal expansion to bring the gas to the final state with the same volume of 3.0 L, and at temperature 110 K. Hint (a) How much work (in J) is done on the gas during the isobaric compression? Wcompression = J (b) How much work (in J) is done on the gas during the isothermal expansion? Wexpansion = J

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, elizabethprasad2
How many grams of n2h4 will be consumed by 23 g of n2o4
Answers: 1

Chemistry, 22.06.2019 09:00, angelrenee2000
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

You know the right answer?
We now consider three moles of ideal gas at the same initial state (3.0 L at 273 K). This time, we w...
Questions in other subjects:



Physics, 01.12.2020 23:30





Advanced Placement (AP), 01.12.2020 23:30

Mathematics, 01.12.2020 23:30

Mathematics, 01.12.2020 23:30

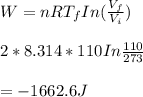

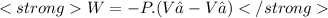
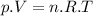
![p= [n.R.T] ÷ V = [3 mol × 0.082 (L.atm÷mol.K) × 273 K] ÷ 3 L](/tpl/images/0676/7490/eb257.png)