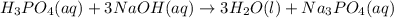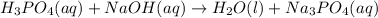Chemistry, 02.06.2020 23:00 angelinamadriga
In a neutralization reaction with a strong acid or base, all of the hydrogens from the acid and the hydroxides from the base react to form water.
Balance the following neutralization reaction:
H3PO4 (aq) + NaOH (aq) â H2O (l) + Na3PO4 (aq)
Enter the appropriate coefficient to the left of the respective term. In the event that your coefficient would be "1" be sure to write "1" as opposed to leaving it blank.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:40, deedee363
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 23.06.2019 03:50, KAITLYN007
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1

Chemistry, 23.06.2019 15:20, masonrochester7
How many stereoisomers will be formed from the addition of phenyllithium to this molecule?
Answers: 1
You know the right answer?
In a neutralization reaction with a strong acid or base, all of the hydrogens from the acid and the...
Questions in other subjects:


Geography, 27.09.2021 09:10

Computers and Technology, 27.09.2021 09:10

History, 27.09.2021 09:10

Mathematics, 27.09.2021 09:10


Mathematics, 27.09.2021 09:20


Business, 27.09.2021 09:20