
Question 17 In the Haber reaction, patented by German chemist Fritz Haber in 1908, dinitrogen gas combines with dihydrogen gas to produce gaseous ammonia. This reaction is now the first step taken to make most of the world's fertilizer. Suppose a chemical engineer studying a new catalyst for the Haber reaction finds that 786. liters per second of dinitrogen are consumed when the reaction is run at 222.°C and 0.35atm. Calculate the rate at which ammonia is being produced. Give your answer in kilograms per second. Round your answer to 2 significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, UaRemomGAY
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3
You know the right answer?
Question 17 In the Haber reaction, patented by German chemist Fritz Haber in 1908, dinitrogen gas co...
Questions in other subjects:

Mathematics, 26.10.2021 02:20

English, 26.10.2021 02:20

Mathematics, 26.10.2021 02:20

Mathematics, 26.10.2021 02:20


Computers and Technology, 26.10.2021 02:20



Mathematics, 26.10.2021 02:20

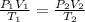
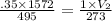
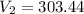 liter .
liter . 

