
Chemistry, 30.05.2020 09:57 liamgreene90
If you start with 6 mol of nitrogen gas (N2+) what mass (g) of ammonia (NH4) will be produced?
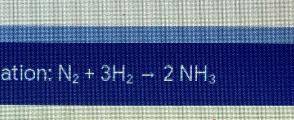

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1

Chemistry, 22.06.2019 19:00, ghernadez
Imagine that a new planet is discovered with two moons of equal mass: moon a and moon b. the mass of the new planet is greater than the combined mass of its moons. moon a is farther away from the new planet than moon b. what is the planet's gravitational pull on moon a compared to the planet's gravitational pull on moon b? the planet's gravity repels moon a with a greater force than it repels moon b, which is why moon a is farther away. the gravitational pull on moon b is greater than on moon a because moon b is closer to the new planet than moon a. the gravitational pull on moon b is greater than on moon a because moon b is farther away from the new planet than moon a. the gravitational pull on moon a is the same as the gravitational pull on moon b because distance does not affect the planet's gravity.
Answers: 1
You know the right answer?
If you start with 6 mol of nitrogen gas (N2+) what mass (g) of ammonia (NH4) will be produced?
Questions in other subjects:


Social Studies, 30.07.2019 07:30

English, 30.07.2019 07:30

History, 30.07.2019 07:30



Computers and Technology, 30.07.2019 07:30





