
Chemistry, 28.05.2020 02:58 cyclonenetwork1188
Consider the following system at equilibrium where H° = 10.4 kJ, and Kc = 1.80×10-2, at 698 K. 2HI(g) H2(g) + I2(g) When 0.40 moles of H2(g) are added to the equilibrium system at constant temperature: The value of Kc The value of Qc Kc. The reaction must run in the forward direction to restablish equilibrium. run in the reverse direction to restablish equilibrium. remain the same. It is already at equilibrium. The concentration of I2 will

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, alexusnicole817
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 07:20, rex1578
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 15:30, 20cschultz
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 22.06.2019 19:10, asdfghhk9805
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
Consider the following system at equilibrium where H° = 10.4 kJ, and Kc = 1.80×10-2, at 698 K. 2HI(g...
Questions in other subjects:



Chemistry, 06.07.2019 08:40



English, 06.07.2019 08:50

Biology, 06.07.2019 08:50

Mathematics, 06.07.2019 08:50


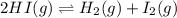
![Qc=\frac{[H_2][I_2]}{[HI]^2}](/tpl/images/0668/1770/280b3.png)


