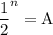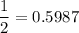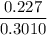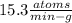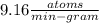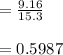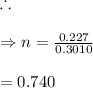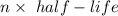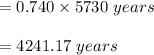In living organisms, C-14 atoms disintegrate at a rate of 15.3 atoms per minute per gram of carbon. A charcoal sample from an archaeological site has a C-14 disintegration rate of 9.16 atoms per minute per gram of carbon. Estimate the age of this sample in years. The half-life of C-14 is 5730 years. (enter only the number of years in standard notation, not the unit years)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:50, ShlomoShekelstein
Why do scientists look for patterns in the world? a. patterns can explain observations. b. patterns never change, no matter what. c. patterns are easy for scientists to detect. d. patterns are all the same, through all time.
Answers: 1


Chemistry, 22.06.2019 12:30, kingbot350
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

You know the right answer?
In living organisms, C-14 atoms disintegrate at a rate of 15.3 atoms per minute per gram of carbon....
Questions in other subjects:

Spanish, 31.10.2019 06:31

Mathematics, 31.10.2019 06:31




Mathematics, 31.10.2019 06:31


Mathematics, 31.10.2019 06:31

Mathematics, 31.10.2019 06:31

Biology, 31.10.2019 06:31

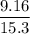 = 0.5987
= 0.5987