

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:10, ellemarshall13
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1


Chemistry, 22.06.2019 21:30, imalexiscv
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
A solution is formed by mixing 15.2 g KOH into
1.200 kg water. Use the periodic table to find...
1.200 kg water. Use the periodic table to find...
Questions in other subjects:

History, 12.12.2020 17:00

Chemistry, 12.12.2020 17:00




History, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Mathematics, 12.12.2020 17:00

Health, 12.12.2020 17:00

 of formula units in that
of formula units in that 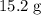 of
of 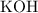 (the solute of this solution.)
(the solute of this solution.) 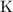 atom, One
atom, One  atom, andOne
atom, andOne  atom.
atom.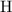 atoms.
atoms. .
.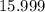 .
. .
. ) is
) is  .
. sample would be:
sample would be: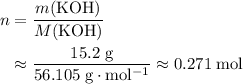 .
.

