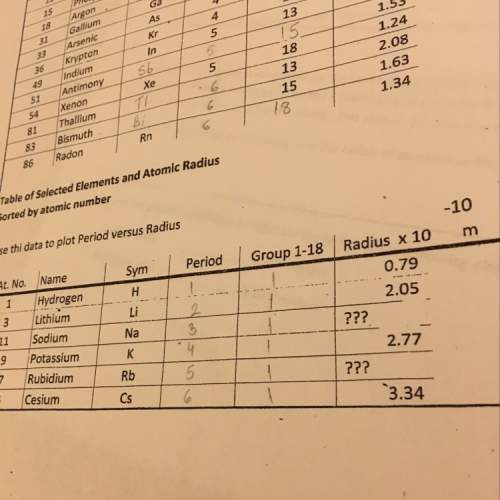
Chemistry, 21.05.2020 23:58 keenonwhite6801
Two liters of oxygen (O2) is collected over water at 20oC. If the total pressure in the container is 95.00 kPa, what is the partial pressure of the oxygen gas?
A) 92.00 kPa
B) 92.67 kPa
C) 95.00 kPa
D) 97.33 kPa
Temperature (oC) Vapor pressure (kPa)
0 0.61
20 2.33
60 19.92
100 101.3

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Averybeam300
Five students had to answer the question how are elements arranged in a periodic tabledamon: i think the elements are arranged by increasing massflo: i think the elements are arranged according to their properties sienna: i think the elements are arranged by when their discovers kyle: i think the elements are arranged according to how common they areglenda: i don't agree with any of themwho is right
Answers: 1


Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 17:30, kiaramccurty
What type of organic molecule comprises the majority of a potato?
Answers: 1
You know the right answer?
Two liters of oxygen (O2) is collected over water at 20oC. If the total pressure in the container is...
Questions in other subjects:

English, 18.12.2020 14:00

Physics, 18.12.2020 14:00

Mathematics, 18.12.2020 14:00


SAT, 18.12.2020 14:00


History, 18.12.2020 14:00

Biology, 18.12.2020 14:00

Mathematics, 18.12.2020 14:00