20 POINTS AND GIVING BRAINLIEST
The following reaction shows the products when sulfuric a...

Chemistry, 22.05.2020 01:05 baileyflemingde
20 POINTS AND GIVING BRAINLIEST
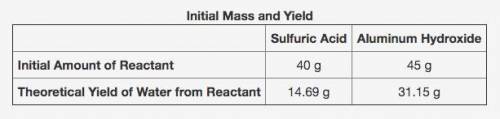
The following reaction shows the products when sulfuric acid and aluminum hydroxide react.
2Al(OH)3 + 3H2SO4 → Al2(SO4)3 + 6H2O
The table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. (See attached image)
What is the approximate amount of the leftover reactant?
A) 20.89 g of sulfuric acid
B) 22.44 g of sulfuric acid
C) 21.22 g of aluminum hydroxide
D) 23.78 g of aluminum hydroxide

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:40, sadcase85
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na, so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 17:30, nijanicole164
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
You know the right answer?
Questions in other subjects:

Mathematics, 28.06.2019 09:30

Biology, 28.06.2019 09:30

Mathematics, 28.06.2019 09:30




Mathematics, 28.06.2019 09:30

Geography, 28.06.2019 09:30

Mathematics, 28.06.2019 09:30

Chemistry, 28.06.2019 09:30



