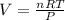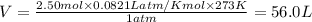The volume, in liters, occupied by 2.50 moles of N2 gas.
calculate at STP...


Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2
You know the right answer?
Questions in other subjects:


History, 28.10.2020 21:50


Mathematics, 28.10.2020 21:50

History, 28.10.2020 21:50

Mathematics, 28.10.2020 21:50

Health, 28.10.2020 21:50


Mathematics, 28.10.2020 21:50

Spanish, 28.10.2020 21:50

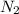 gas at STP is 56.0L
gas at STP is 56.0L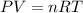
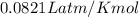
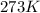 (at STP)
(at STP)