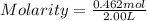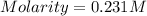What is the molarity if 2.00 liters containing 49.0 grams of sodium carbonate [Na2CO3)?
(Molar...

Chemistry, 21.05.2020 00:12 lorenzo1beaton
What is the molarity if 2.00 liters containing 49.0 grams of sodium carbonate [Na2CO3)?
(Molar mass of Na is 22.99 g/mol, C is 12.01 g/mol and O is 16.00 g/mol.)

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, AaronEarlMerringer
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1

Chemistry, 23.06.2019 02:50, agm0102
What is the typical rotational frequency frot for a molecule like n2 at room temperature (25∘c)? assume that d for this molecule is 1å=10−10m. take the total mass of an n2 molecule to be mn2=4.65×10−26kg. you will need to account for rotations around two axes (not just one) to find the correct frequency. express frot numerically in hertz, to three significant figures.
Answers: 3

Chemistry, 23.06.2019 04:20, monifaWilson
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2
You know the right answer?
Questions in other subjects:




Physics, 15.10.2020 09:01


Mathematics, 15.10.2020 09:01

History, 15.10.2020 09:01




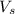 = volume of solution in L
= volume of solution in L =
=