
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. An industrial chemist studying this reaction fills a 500. mL flask with 3.7 atm of sulfur dioxide gas and 2.3 atm of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of sulfur trioxide gas to be 2.2 atm. Calculate the pressure equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. Round your answer to 2 significant digits.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, carsonjohnsonn
If a 12-v battery is connected to a circuit that has a current of 3.0 a, what is the total resistance in the circuit? 36 ohms 4 ohms 0.25 ohms
Answers: 1


Chemistry, 23.06.2019 05:20, cjking2320
Explain how global warming could have affected yellowstone frog and salamander habitat's, resulting in changes in the populations of these species
Answers: 2
You know the right answer?
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid...
Questions in other subjects:

Mathematics, 10.02.2021 04:00

Mathematics, 10.02.2021 04:00

Chemistry, 10.02.2021 04:00

Social Studies, 10.02.2021 04:00

Mathematics, 10.02.2021 04:00

Mathematics, 10.02.2021 04:00




English, 10.02.2021 04:00

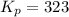
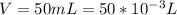
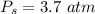
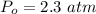
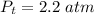
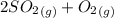 ⇄
⇄ 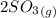
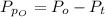
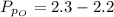
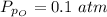
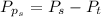
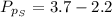
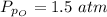
![K_p = \frac{[P_t]^2}{[P_p__{o}} ]^2 [P_p__{s}}]}](/tpl/images/0656/7414/8f8eb.png)
![K_p = \frac{[2.2]^2}{[ 0.1 ]^2 [{ 1.5}]}](/tpl/images/0656/7414/2f87d.png)


