
Chemistry, 19.05.2020 17:00 dontcareanyonemo
A student wishes to calculate the experimental value of Ksp for AgI. S/he follows the procedure in Part 3 and finds Ecell to be 0.839 V. S/he does not perform Part 2; instead, s/he uses the theoretical slope of the Nernst plot for an Ag/Ag+ concentration cell, as instructed by her/his TA; this value is −0.0591 V. (a) What is the concentration of Ag+(aq) in the saturated solution of AgI (i. e., [Ag+ ]dilute)? ([Ag+ ]conc = 1.0 ✕ 10−1 M.) M (b) Using [I− ] = 0.20 M, calculate the experimental Ksp. (c) Suppose s/he mistakenly uses 1.039 V as Ecell. How does this affect [Ag+ ]dilute? Will it be too high, too low, or unaffected?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:00, smelcher3900
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2

Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1


Chemistry, 23.06.2019 09:30, Dariana123
Which of the following is not a characteristic of a hydrogen bond? 1. it is responsible for the unusual physical properties of water. 2. it is weaker than a covalent bond. 3. it is stronger than other dipole-dipole interactions. 4. it can occur when hydrogen is covalently bound to very electronegative elements liks f, cl, br and i.
Answers: 1
You know the right answer?
A student wishes to calculate the experimental value of Ksp for AgI. S/he follows the procedure in P...
Questions in other subjects:


Advanced Placement (AP), 25.11.2021 05:50


Social Studies, 25.11.2021 05:50

Mathematics, 25.11.2021 05:50





 × 1.0 × 10⁻¹ M
× 1.0 × 10⁻¹ M
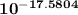 × 1.0 × 10⁻¹ M
× 1.0 × 10⁻¹ M



