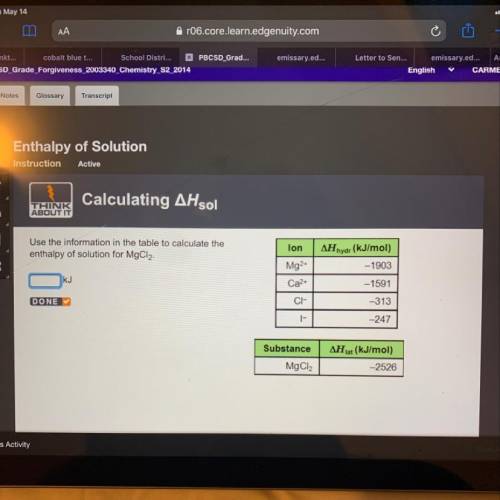
Use the information in the table to calculate the enthalpy of solution for MgCl2. KJ
...


Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 18:00, heggestade
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3


Chemistry, 23.06.2019 00:30, vane6176
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3

Chemistry, 23.06.2019 01:00, skatelife8974
What is the chemical name of the compound ti2o3?
Answers: 2
You know the right answer?
Questions in other subjects:




English, 14.11.2019 11:31

History, 14.11.2019 11:31

English, 14.11.2019 11:31

Mathematics, 14.11.2019 11:31

English, 14.11.2019 11:31