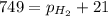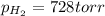Chemistry, 06.05.2020 17:08 22katelynfrankouqqrb
As the magnesium reacts, the hydrogen gas produced is collected by water displacement at 23.0oC. The pressure of the gas in the collection tube is measured to be 749 torr. Given that the equilibrium vapor pressure of water is 21 torr at 23.0oC, calculate the pressure that the H2(g) produced in the reaction would have if it were dry.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, 2024daisjavien
Which actions would increase the rate at salt dissolves in water? stir the water? crush the salt? use less water? heat the water? cool the salt
Answers: 3



Chemistry, 22.06.2019 22:30, brianna5626
How do limiting factors most affect population size? ostop population growthrestrict population growthincrease population sizeresult in positive impactso
Answers: 1
You know the right answer?
As the magnesium reacts, the hydrogen gas produced is collected by water displacement at 23.0oC. The...
Questions in other subjects:


Physics, 09.03.2021 21:30



Chemistry, 09.03.2021 21:30

English, 09.03.2021 21:30

Mathematics, 09.03.2021 21:30



Mathematics, 09.03.2021 21:30

 produced in the reaction would have if it were dry will be 728 torr
produced in the reaction would have if it were dry will be 728 torr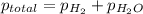
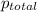 =total pressure of gas = 749 torr
=total pressure of gas = 749 torr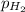 = partial pressure of hydrogen = ?
= partial pressure of hydrogen = ?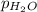 = partial pressure of water = 21 torr
= partial pressure of water = 21 torr