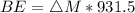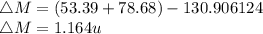Iodine-131 is a radioactive isotope that is used in the treatment of cancer of the thyroid. The natural tendency of the thyroid to take up iodine creates a pathway for which radiation (β− and γ) that is emitted from this unstable nucleus can be directed onto the cancerous tumor with very little collateral damage to surrounding healthy tissue. Another advantage of the isotope is its relatively short half-life (8 days). Calculate the binding energy of iodine-131 and the binding energy per nucleon. The mass of iodine-131 is 130.906124 u.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, micvar9646
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 03:50, sgslayerkingminecraf
Which of the following statements about acidic water is true? a. acid has no effect on the h, o molecules. b. the solution contains a larger number of oh ions than h, o ions. c. the solution contains a larger number of h, o ions than qh ions. d. the solution contains an equal number of h, o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 09:10, cheesedoodle
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1
You know the right answer?
Iodine-131 is a radioactive isotope that is used in the treatment of cancer of the thyroid. The natu...
Questions in other subjects:



Mathematics, 06.03.2020 23:04

Mathematics, 06.03.2020 23:04


Mathematics, 06.03.2020 23:04

Mathematics, 06.03.2020 23:04

Mathematics, 06.03.2020 23:04

Mathematics, 06.03.2020 23:04

Mathematics, 06.03.2020 23:04