Chemistry, 05.05.2020 17:37 canyonmorlan
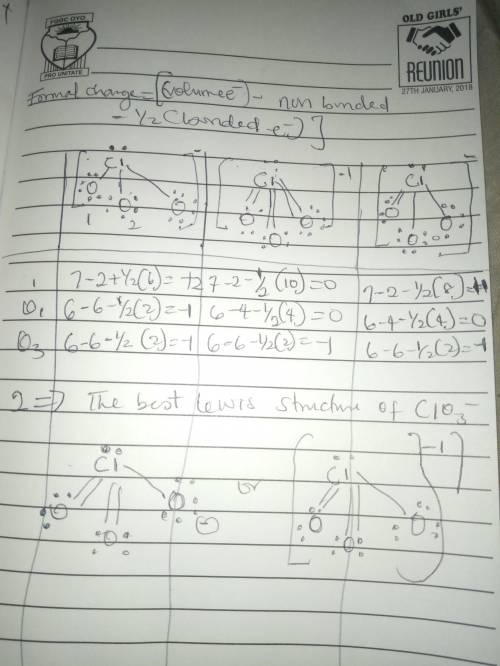
The formal charge is the "charge" an element would have in a molecule or ion if all of the bonding electrons were shared equally between atoms. We can draw three inequivalent Lewis structures for the thiocyanate ion , SCN- . The concepts of formal charge and electronegativity can help us choose the structure that is the best representation.1. Assign formal charges to the elements in each of the structures below. ABCFormal ChargeSCN-12010-100-12. The best Lewis structure for SCN- is .

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, marcusajns
Y=‐1x + 7 if y has a value of ‐24 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 15:00, tcapele252
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 18:00, kamjay2006
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1

Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
The formal charge is the "charge" an element would have in a molecule or ion if all of the bonding e...
Questions in other subjects:




Mathematics, 21.07.2020 07:01


Chemistry, 21.07.2020 07:01

Chemistry, 21.07.2020 07:01



Mathematics, 21.07.2020 07:01