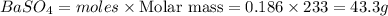Chemistry, 13.01.2020 23:31 bullsfan4584
Calculate the mass of the precipitate formed when 2.27 l of 0.0820 m ba(oh)2 are mixed with 3.06 l of 0.0664 m na2so4.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 06:30, 91miketaylor
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 12:00, carvajalj2520
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 13:00, cnfndbxbfbdb2031
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
You know the right answer?
Calculate the mass of the precipitate formed when 2.27 l of 0.0820 m ba(oh)2 are mixed with 3.06 l o...
Questions in other subjects:


Physics, 19.11.2020 20:10



Arts, 19.11.2020 20:10




Arts, 19.11.2020 20:10

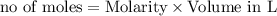
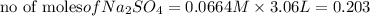
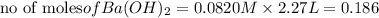
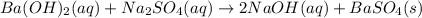
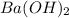 reacts with 1 mole of
reacts with 1 mole of 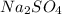
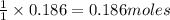 of
of 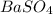 precipitate
precipitate